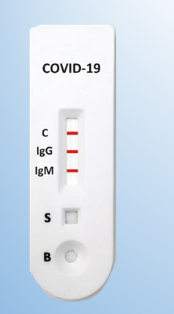
What is Abbott rapid test?
The Panbio rapid or simple home test is an easy to use test that allows prompt results in just a couple of minutes. The main advantage of the Abbott rapid tests is that they are easy to use they don’t require any special equipment and training.
One type of rapid test is for example pregnancy test or the test for measuring of glucose levels performed by millions of people with diabetes. The Covid test is performed on the same very small volume of blood and the Panbio test contains lancettes to take the blood sampling.
Panbio Rapid diagnostic tests (RDTs) could accelerate the etiologic diagnosis of illnesses in a number of cases (e.g., sepsis, respiratory tract infections, unsymptomatic Covid and meningitis).
Interpretation of now available RDTs isn’t necessarily simple, and so, they can’t yet replace traditional PCR tests. Institutional priorities, integration with lab competencies, and cost are issues which need to be taken into consideration when choosing RDTs to execute locally. RDTs can enhance antimicrobial prescribing at minimal level when the antimicrobial stewardship group is involved; nonetheless, this should be tracked. Accessible biomarkers for disease detection, chiefly C-reactive protein (CRP) and procalcitonin (PCT), are helpful, when implemented appropriately, to decrease antibiotic exposure in both outpatient and inpatient setting, particularly for severe respiratory tract infections
What are the main advantages of performing Simple Panbio tests?
• Tests based on agglutination, immuno-dot, immuno-chromatographic and/or immuno-filtration techniques with high quality, easy-to-use tests for use in resource poor settings.
• Quick And simple to do — 10 minutes to two hours and need little if any extra equipment.
• Are Made to be used with person or a restricted number of samples, making them cheaper than ELISAs in non throughput labs.
• Possibility to store room temperature for an extended time period.
• Same-day Results give timely therapy interventions.
Principe of Abbott Panbio COVID-19 Cassette Rapid test?
The 2019-nCoV IgG/IgM Rapid Test Cassette from Abbott (Whole Blood/Serum/Plasma) is a qualitative membrane-based immunoassay for the detection of IgG and IgM antibodies to 2019-nCoV in whole blood, plasma or serum specimen. This test contains two components, an IgG element and an IgM component. From the IgG part, anti-human IgG is coated in IgG test line region. Throughout testing, the specimen reacts with 2019-nCoV antigen-coated particles in the test cassette. The mixture then migrates up to the membrane chromatographically by capillary action and reacts with the anti-human IgG in IgG test line region, if the specimen includes IgG antibodies to 2019-nCoV. A colored line will look in IgG test line region as a result of this. In the same way, anti-human IgM is coated in IgM test line region and when specimen contains IgM antibodies to 2019-nCoV, the conjugate-specimen complex reacts with anti-human IgM. A colored line appears in IgM test line region as a result. Therefore, if the specimen includes 2019-nCoV IgG antibodies, a colored line will appear at IgG test line area. If the specimen includes 2019-nCoV IgM antibodies, a colored line will appear in IgM test line region. If the specimen doesn’t contain 2019-nCoV antibodies, no colored line will look in both of the test line areas, indicating a negative outcome.
How to perform a rapid test for COVID-19?
Note of caution: Panbio Rapid tests for any infectious diseases should only be performed in a diagnostic laboratory by authorized personal, wearing protection.
Allow the test, specimen, buffer or controls to reach room temperature (15-30°C) Before to testing.
1. Eliminate the test cassette from the foil pouch and use it within a hour. Best results will be
Obtained if the test is done immediately after opening the foil pouch.
2. Put the tape on a clean and level surface.
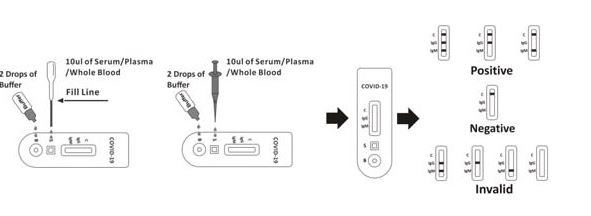
For Serum or Plasma specimen:
Тo utilize a dropper: Hold the dropper vertically, draw on the specimen to the fill line
(roughly 10uL), and then move the specimen to the specimen well (S), then add two drops
Of buffer (approximately 80 uL), and start the timer.
Of buffer (roughly 80 uL), and start the timer
• To use a dropper: Hold the dropper vertically, draw the specimen around 1 cm over the fill
Line and move 1 full drop (approx. 20μL) of specimen to the sample well(S). Then add two
Drops of buffer (approximately 80 uL) and start the timer.
• To utilize a pipette: To transfer 20 uL of whole blood to the specimen well(S), then add two
Drops of buffer (approximately 80 uL), and start the timer
• To use a dropper: Hold the dropper vertically, draw the specimen around 1 cm above the fill
Line and transfer 1 full fall (approx. 20μL) of specimen into the sample well(S). Then add 2
Drops of buffer (approximately 80 uL) and then start the timer.
• To utilize a capillary tube: Fill the capillary tube and transfer approximately 20uL of
Fingerstick whole blood flow to the specimen well (S) of the test tape, then add two
Drops of buffer (roughly 80 uL) and start the timer. See illustration below.
3. Await the colored line(s) to look. Read results in 10 minutes. Do not interpret the Outcome
Following 20 minutes.
Note: It is suggested to not use the buffer, past 6 months after opening the vial.
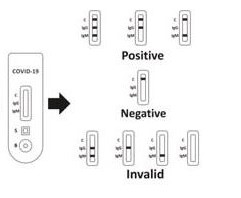
How to interpret the Panbio results?
IgG POSITIVE:* Two colored lines appear. One colored line should always appear in the controller
Line area (C) and another line should be in the IgG line up area.
IgM POSITIVE:* Two colored lines appear. Line area (C) and another line should be in the IgM line area.
IgG and IgM POSITIVE:* Three colored lines appear. One colored line should appear in The controller Line region (C) and 2 test lines ought to maintain the IgG line area and IgM line area.
*NOTE: The Intensity of this color in the test line areas may fluctuate depending on the concentration of 2019-nCoV antibodies within the specimen. Therefore, any shade of color in the test line Area Should be considered favorable.
NEGATIVE: One colored line appears in the control line region (C). No line appears in the IgG Region and IgM region.
INVALID: Control line fails to appear. Insufficient specimen volume or incorrect procedural techniques Review the procedure and repeat the test a new evaluation. If the problem persists, discontinue using the test kit immediately and contact Your local distributor. Internal Procedural controls are contained in the test. A colored line appearing at the control regioni nternal procedural control. It confirms sufficient specimen volume and correct procedural technique. Control standards are not supplied with this kitNonetheless, it is recommended that positive and negative controls be tested as a PCR laboratory practice to confirm the test. Process to confirm proper test performance.
What are the limitations of Simple Panbio Cassette Test for SARS-CoV-2 ?
1. The 2019-nCoV IgG/IgM Rapid Test Cassette (Whole Blood/Serum/Plasma) is for in vitro diagnostic use only. This Abbott evaluation test should be used for detection of IgG and IgM antibody to 2019-NCoV in whole blood, serum or plasma specimens. Neither the quantitative value nor the speed this qualitative evaluation.
2. The 2019-nCoV IgG/IgM Quick Test Cassette (Complete blood/Serum/Plasma) will only indicate
The existence of IgG and IgM antibodies to 2019-nCoV from the specimen and shouldn’t be used
as the only criteria for the identification of 2019-nCoV infections.
3. As with all diagnostic tests, all results must be considered together with other clinical information available to the doctor.
4. If the test result is negative and clinical symptoms persist, further follow-up testing using
Other clinical methods is suggested. A negative result at any moment does not preclude the
possibility of 2019-nCoV disease.
5. The hematocrit level of the whole blood can influence the test outcome.
Between 25% and 65% to get accurate results.
6. The test will reveal negative consequences under the following conditions: The titer of the novel
Coronavirus antibodies in the sample is lower than the minimal detection limit of this test, or
The novel coronavirus antibody has not appeared in the time of sample collection.
Other Covid tests are :
| Accu-Tell Ag Rapid Test | 20 Tests/Kit | € 190.00 | 95.00% | 99.20% | Nasopahryngeal |
| Accu-Tell IgG/IgM Rapid Test | 25 Tests/Kit | € 119.00 | 91.40% | 99.80% | |
| Biocredit Ag Rapid Test | 20 Tests/Kit | € 150.00 | 85.70% | 98.90% | Nasopharynx / Nasopharyngeal |
| ElabScience Ag Rapid Test | 25 Tests/Kit | € 100.00 | 90.32% | 99.21% | Oropharyngeal / Nasal / Nasopharyngeal |
| ElabScience IgG/IgM Rapid Test | 20 Tests/Kit | € 155.00 | 98.81% | 98.02% | |
| Green Spring Ag Rapid Test | 25 Tests/Kit | € 145.00 | 97.14% | 100.00% | Nasal |
| Invbio Saliva Test | 20 Tests/Kit | € 150.00 | 97.10% | 100.00% | |
| Lepu Ag Rapid Test | 25 Tests/Kit | € 175.00 | 92.00% | 99.26% | Nasal |
| Panbio Ag Rapid Test | 25 Tests/Kit | € 155.00 | 91.40% | 99.80% | Nasopahryngeal |
| Panbio IgG/IgM Rapid Test | 25 Tests/Kit | € 220.00 | 96,20% | 100,00% | |
| Tigsun Covid-19 & Flu A/B Test | 25 Tests/Kit | € 218.00 | / | / | |
| Tigsun Covid-19, RSV & Flu A/B Test | 25 Tests/Kit | € 250.00 | / | / | |
| Tigsun Saliva Test | 25 Tests/Kit | € 187.00 | / | / | |
| UnScience Ag Rapid Test | 25 Tests/Kit | € 100.00 | 90.32% | 99.21% | Oropharyngeal / Nasal / Nasopharyngeal |
| UnScience IgG/IgM Rapid Test | 20 Tests/Kit | € 155.00 | 98.81% | 98.02% | |
| VivaDiag Ag Rapid Test | 25 Tests/Kit | € 185.00 | 90.90% | 100.00% | Oropharyngeal / Nasal / Nasopharyngeal |