Introduction
The enzyme-linked immunosorbent assay (ELISA) is a labelled immunoassay that is considered the gold standard of immunoassays. This highly sensitive immunological test is used to detect and quantify substances, including antibodies, antigens, proteins, glycoproteins, and hormones. Detection of these products is achieved by the formation of complexes of antibodies and antigens to produce a measurable result. An antibody is a type of protein produced by an individual’s immune system.
This type of protein has specific regions that bind to antigens. An antigen is a protein that can come from some foreign source and, when bound to an antibody, induces a cascade of events through the body’s immune system. This interaction is used in ELISA tests and allows specific protein antibodies and antigens to be identified with only small amounts of a test sample. PCR-og-ELISA-kits are used to diagnose HIV infection, pregnancy tests, and blood type, among others. This article will discuss the basic principles, procedures, and clinical significance of ELISA.
Aetiology and Epidemiology
Direct ELISA was invented simultaneously by two different research teams by scientists Eva Engvall and Peter Perlman and by Van Weemen and Schuurs. The ELISA was developed by modifying the radioimmunoassay (RIA). This was done by conjugating the labelled antigen and antibody with enzymes instead of radioactive iodine 125. The new method was first used by determining IgG levels in rabbit serum. In the same year, scientists were able to quantify human chorionic gonadotropin in urine by using horseradish peroxidase. Since then, the ELISA method has been used in many different applications and has become a routine diagnostic and laboratory research method throughout the world.
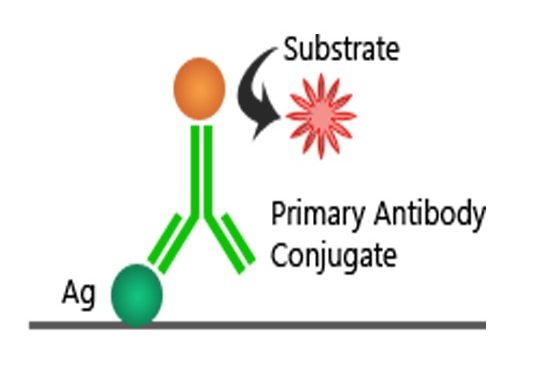
The first ELISA methodology involved substrates and chromogenic reporter molecules in the generation of an observable colour change that monitors the presence of antigen. Further advancement in the ELISA technique leads to the development of electrochemiluminescent, quantitative PCR, and fluorogenic indicators to generate signals. However, some of these techniques are not based on the use of enzyme-linked substrates but on non-enzymatic indicators using the ELISA principle.
The latest development, in 2012, was an enzyme-based ultrasensitive ELISA that manipulates nanoparticles as chromogenic reporters. This technique can generate a colour signal visible to the naked eye, with blue for positive results and red for negative results. However, this method is qualitative and can determine only the presence or absence of an analyte and not its concentration.
Sample requirements and procedure
ELISAs are performed in polystyrene plates, usually, 96-well plates coated to bind proteins very strongly. Depending on the type of ELISA, the test requires a primary and/or secondary detection antibody, analyte/antigen, coating antibody/antigen, buffer, wash, and substrate/chromogen. The primary detection antibody is a specific antibody that binds only to the protein of interest, whereas a secondary detection antibody is an enzyme-conjugated second antibody that binds to a non-enzyme-conjugated primary antibody.
There are four main general steps in completing an ELISA immunoassay. These steps are:
- Coating (with antigen or antibody)
- Blocking (typically with the addition of bovine serum albumin [BSA])
- Detection
- last reading
Detection is carried out by adding a substrate that can generate a colour. There are many substrates available for use in ELISA detection. However, the most widely used are horseradish peroxidase (HRP) and alkaline phosphatase (ALP). The substrate for HRP is hydrogen peroxide which results in a blue colour change. ALP measures the yellow colour of nitrophenol after room temperature incubation periods of 15 to 30 minutes and typically uses p-nitrophenyl phosphate (pNPP) as a substrate.
Diagnostic tests
Enzyme-linked immunosorbent assays are used in many diagnostic tests.[1][2][3] Some of the uses of ELISA may include the following:
Detect and measure the presence of antibodies in the blood
- Autoantibodies (anti-dsDNA, anti-dsg1, ANA, etc.)
- Antibodies against infectious diseases (antibacterial, antiviral, antifungal)
- Hepatitis A, B, C, HIV, etc.
Detect and estimate the levels of tumour markers
- Prostate-Specific Antigen (PSA)
- Carcinoembryonic antigen (CEA)
Detect and Estimate Hormone Levels
- Luteinizing hormone
- follicle-stimulating hormone
- prolactin
- testosterone
- Human chorionic gonadotropin (hCG)
Disease Outbreak Tracking
- Anger
- HIV
- Influenza
Detection of past exposures
- HIV
- Lyme’s disease
- Hepatitis
Screening of donated blood for potential viral contaminants
- anti-HIV-1/2
- anti-HCV
- HBsAg
Drug Abuse Screening
- Amphetamine,
- methamphetamine
- 3,4-methylenedioxymethamphetamine
- Cocaine
- Benzoylecgonine
Interfering factors
Factors that can interfere with proper ELISA testing can occur at any stage of the testing process, beginning with sample collection. The quality and integrity of the assay plate, coating buffer, capture antibody, blocking buffer, target antigen, detection antibody, enzyme conjugate, washes, substrate, and detection signals may interfere with proper ELISA tests. Some of the factors that can interfere with the tests are the following:
- Plate assay: the shape and quality of the wells, the material of the plate, potential pre-activation, uniform or uneven coating
- Buffer: pH, contamination
- Capture and detection antibody: incubation time, temperature, specificity, titer, affinity
- Blocking buffer: cross-reactivity, concentration, contamination
- Target antigen: conformation, stability, epitopes
- Enzyme conjugate: type, concentration, function, cross-reactivity
- Flushes: contamination, frequency, volume, duration, composition
- Substrate: quality/manufacturer
- Detection: instrument-dependent factors
- Reader/human error
Results, reports, critical findings
The data collected from ELISA tests can be quantitative, qualitative or semi-quantitative. Quantitative concentration results are plotted and compared to a standard curve. Qualitative results confirm or deny the presence of a particular antigen/antibody in a sample. Semi-quantitative results compare the intensity of signals, which can compare the relative levels of antigen in a sample.
Once the assay colour changes are measured, the results are plotted on paper or software. Typically, the graph compares optical density to log concentration, which gives a sigmoidal curve. Known concentrations give the standard curve of the plot, and unknowns can then be measured when the sample values are compared to the linear portion of the plotted standard curve.
